Compounds like Kasenite are usually based on yellow potassium ferrocyanide + Carbon source , they are sprinkled on the heated part and be give a short soak time. these will usually produce only a very very shallow depth of carburized material on the surface to increase wear resistance and do not add in the structural stability.
With Pack carburizing a far greater depth of carbon penetration can be achieved. And a hardened "shell" of 1-1,5mm carburized material on an arrowhead will also add in its stability on impact.
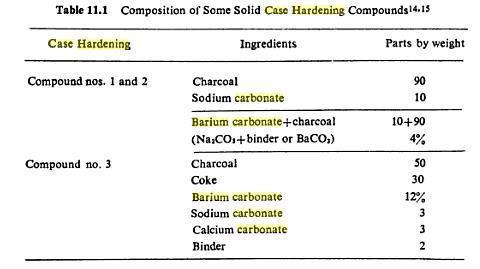
Charcoal alone will work , as will leather scraps but now adays normally a additional catalyst is added to help the carbon mirgrating. The catalyst is usually Barium carbonate plus a bit of sodium carbonate and calcium carbonate.
Sodium carbonate alone as a catalyst will work but can sometimes produce pitting on the steel especially if too much is used .
Barium carbonate may be hard to get and is also classified toxic but sodium carbonate should be easily avaliable (washing soda) and is not toxic. Calcium carbonate can also be easily had from ground seashells, limestone, marble etc.
finely gound charcoal mixed with the catalyst and formed to small pellets witha binder like oil or tar works better than large lumps of charcoal.
There are many recipes out there, just do a google search for "case hardening and barium carbonate" or case hardening sodium carbonate". A starting point would be about 10% catalyst 90% charcoal. But some recipes go up as far as 40% of catalyst. Well, you got to experiment a little, since it also depends on the steel, time and temperature you use.
Before I would resort to pack carburizing I would try to use a faster Quenchant fot your steel. Depending on the carbon content a very fast quenchant is able to harden steels that would not harden im Water normally.
If the carbon content of your mild steel is very very low it may not work but its said that super quench is able to harden steels like 1020 (0,2%C) which you cant otherwise harden in water or oil.
If you also cannot get the ingredients for the super quench there is still the posibility to use a 20% concentrated sodium hydroxide solution (lye) as a quenchant, but this was banned from use in the industry for some reasons because it is dangerous, do not inhale the steam/fumes and if you get some lye in your eyes you can go blind easily. So do this only as last resort at your own risk.!!

Before the Bessemer process made it feasible to effectively control the amount of carbon in steel, blacksmiths generally had only iron or tool steel to work with. The Bessemer process gave the steel manufacturers the ability to produce steel in a variety of carbon levels. Mild steel (1005, 1018, and the like) was touted as the all purpose steel destined to replace wrought iron. The manufacturers claimed that it was also suitable for many tools, but that it should be quenched in a solution of sodium hydroxide.
At Sandia Labs, Robb and his cohorts experimented with this lye quench and, a bit to their surprise, they found that mild steel hardened considerably more that expected. Metallurgists and others will tell you quite readily that mild steel won't harden. It may get a little harder than if annealed, however it doesn't harden in the typical toolmaker's sense of hardening. Generally speaking, in a plain water quench you shouldn't expect to get more than Rockwell ratings in the low to mid 30's. Robb found that the sodium hydroxide quench resulted in average Rockwell ratings in the 43-45 range, with an occasional test result as high as 48.
So, Robb started using this solution at Sandia Labs, but installed a vented hood system over the quench tank. This stuff is pretty harsh and the need for a vented hood was a no-brainer.
Then OSHA arrived on the scene and insisted that the use of the sodium hydroxide solution cease. The result was that Robb and the Sandia Labs metallurgical lab crew went to work to find a replacement solution. It had to give hardness results comparable to the sodium hydroxide solution, and it should be bio-degradable if possible. The result of their experimentation was what is now generally referred to as Gunter's Super Quench. The formulation is as follows:
• 5 gallons of water (This a good volume to work with for quenching, and there are plenty of buckets and pails around just the right size.)
• 5 lbs table salt (plain or iodized, canning salt or rock salt, it makes no difference.)
• 32 oz Dawn Liquid Dishwashing Detergent Blue. 28 oz if the label says Concentrated.(Blue was chosen because that's what happened to be available at the moment. It was noted later on that as the solution deteriorated to the point that it should be disposed of, the color slowly changed to green. Hence, the blue detergent is recommended. Any other blue colored liquid detergent could work just as good.)
• 8 oz Shaklee Basic I. (The solution needs a surfactant to maximize contact between the solution and the piece being quenched. Amway Basic H will also work. Your local farmer's supply should be able to help here, as similar surfactants are used to facilitate the distribution of fertilizer in soil. In response to a question from the viewing stands, Robb said that just about any wetting agent should do, even the stuff photo film developers use. Just follow directions on amount of agent to be added to a given amount of water, then scale up or down to the 5 gallons of water used in this formulation.)
Heat your iron to 1550 degrees Fahrenheit, and quench. No tempering is needed.
So, he mixed up a batch right there in front of us and used it for his next demonstration. He took a piece of 1/2" 1018 and cut off a piece about 3" long. This piece was heated in the gas forge, and a cold chisel end was forged on to it. Robb heated the piece to 1550 (critical temp for mild steel), and quenched it in the solution. He then took his new "chisel" and proceeded to use it to cut almost through the parent bar. Then, he did it again. The cut bar and the chisel were passed around for all to take a good look at. The edge on the chisel was not deformed in any way. The top had not mushroomed, nor did it even show any evidence of having been hit with the hammer. But, so as not to mislead us, Robb said that a chisel of this type might be good for 7 or 8 cuts maximum. He recommends this quench for tools such as spring fullers and many treadle hammer tools/dies/fullers. He showed, and used, one such spring fuller that is made of mild steel and quenched in the solution. He has been using this particular fuller for several years with no ill effect. The fuller is unmarred, and the spring is still strong.
I was impressed, to say the least. I was not the only person there who was "wowed" by this little "trick". Robb said the quench is good for anything up to 50 points of carbon. Above this carbon level this quench should not be used.










 Private messages
Private messages