I hope you guys were reading up on the subject outside the forum

....anyways, I did.
 Paris Green
Paris Green....quite a history on that one.

Turns out it's a Coppper/Arsenic compound that was mostly used as a rodenticide/insecticide due to it's wonderful toxicity (
"...once was used in Parisian sewers to kill rats, hence the common name..."), but despite that, it was also used as a pigment in paints and inks for it's vibrant blush green tint.
Now I can somewhat understand why it was mentioned in the fabric/cordage treatment solution (and not necessarily as a pigment or a waterproofing agent). As the book said, it was used in very small amounts and was basically there to prevent the clothing from being infested with insects (
I'm guessing lice, fleas, and other parasites...as well as to prevent the clothing from being chewed up by rats - perhaps while in storage). Not sure if it was a good fungicide/preservative (
as that would be a good property in the waterproofing solution). - Here's the
"Wiki"Viridian is a bit different, though also green, it's actually a Chromium compound and is primarily used as a pigment. -
"Wiki"
Also it's interesting that you mentioned Sailing ships, because that really rang a bell -
Vikings!!! 
There is a whole article on the reconstruction of their longboats that had wool sails. And there is much said about fabrication and preparation, a good part of whic is...you guessed it - waterproofing and tensile strenth.
"PDF Link" 
...pretty cool stuff actually.
Here is a quick cutout...
...
Air permeability and smo¨rring (dressing)
During sail fabric testing at UMIST in an attempt
to understand the functioning of wool sails the
decision was made to test air permeability. The
Shirley Air Permeability Tester was used on the
Skuldelev 1 replica fabric and on other similar test
fabrics. Initial tests showed that the fabrics had
such high permeability that the standard pressure
drop of 10 mm water pressure could not be
achieved at the highest flow rate. The test was
modified to 2 mm water pressure before readings
could be taken that were typically between 100–
150 cc/sec flow rate. Fabrics with such high permeability
would not function as sails because air
would flow through the sail and eliminate the
pressure difference between the front and rear that
creates the draw. It had been suggested that half
Figure 3. Schematic diagram of the Otar square sail. (Drawing:
Viking Ship Museum)
NAUTICAL ARCHAEOLOGY, 31.2
208
fulling the fabrics would reduce the permeability
but tests on loom-state and fulled fabrics showed
that air permeability was typically only reduced
by 30–35% by fulling, which was not enough to
resolve the problem.
A fragment of the Nordmore sail was then
tested under the same conditions and was found
to have much lower air permeability, 10–20 cc/sec.
The Nordmore sail had been impregnated with a
resin like finish and this tied-in with comments in
the literature about smo¨ rring (Andersen, 1995).
Smo¨ rring involves a two-stage process of, firstly,
brushing into the fabric an emulsion of water,
horse fat, (from beneath the mane) and ochre.
This is allowed to dry and then hot liquid beef
tallow is rubbed and smoothed into the sail. This
process was used twice to improve the light
stretchy sail of the Roar Ege and was also applied
to the Sif Ege sail (Andersen, pers. comm.). To
establish the effect of smo¨ rring, the Skuldelev 1
fabric and the other test webs were tested before
and after treatment and as part of this experiment
the extension of a 5 cm strip at 100 N loading was
also measured before and after. The air permeability
tests show that smo¨ rring dramatically
reduces the airflow. The sample SK1/4,
(Skuldulev 1 oppsett 4) shows that a high air
permeability of 111 cm/sec can be ‘tuned’ down to
4 cm/sec by the two stage process of horse fat and
ochre followed by beef tallow. The results for
Skuldelev 1 oppset 2 and Skuldelev 1 oppset 5,
both show the effect of each of the two stages. The
first application lowered the air flow from 68–
85 cc/sec down to 18 cc/sec, and the beef tallow
application reduced it further, to close to zero if
required, dependant on the quantity of beef tallow
applied. The other interesting result is that the
three warp-weighted loom fabrics included in this
test, AN1, AN2 and BF all give air flow values in
the region of 30 cc/sec prior to smo¨ rring and this
suggests that it is easier for a woman of average
strength to produce a very tight fabric on a
warp-weighted loom as compared with the horizontal
loom fabrics which all have initial air flow
values above 62 cc/sec, (62–111).
Finally the 5 cm strip testing demonstrated that
smo¨ rring reduced the low load (100 N/5 cm) extension
by approximately 30–40%, thus increasing
the tensile modulus accordingly. Smo¨ rring can
therefore be seen as an important aid in the
successful use of coarse woollen sails. It enables
the air permeability to be reduced to a level at
which the sail will develop a good draw and it also
has the effect of trimming the elasticity of the sail.
The inclusion of ochre in the mix is also of
significance. It undoubtedly acts as a good filler
inhibiting air flow by filling the voids in the fabric,
but it also has anti-bacterial properties which help
to prevent the wet sail from rotting.
Conclusions
The experimental archaeology associated with the
reconstruction of woollen sails for the reconstructed
Skuldelev ships has radically altered the
understanding of the functioning and efficiency of
such sails. In 1975 Svend Larsen concluded his
book, Vikingsernes hav, by stating that beating to
windward cannot be done with woollen sails
because they are ‘fleecy, nappy, yielding and
leaky’ (Larsen, 1975). It is now known that high
cover factor woollen square-sails could beat at 66�
into the wind and most likely out-perform linen
and hemp sails. Furthermore it has been proved
conclusively that the process of smo¨ rring enables
the properties of the wool sail to be improved and
‘trimmed’ during use.
In a small way the research carried out at
UMIST as part of the Raphael ‘Seafaring’ project
has added to the understanding derived from
experimental archaeology by providing comparative
quantitative data within a framework of the
fibre physics and fabric mechanics essential for
the evaluation of the results. It is to be hoped that
this work will help to provide the foundation for a
better understanding of the design and performance
of woollen square-sails together with a
clearer appreciation of the high level of technical
skills and knowledge developed empirically by our
forebears.
And last - one of my favorites since I've dealt with it quite a bit when trying to make some homebrew metal casting investment - Diatomite (
Diatomaceous earth) mentioned in a few recipes...
The Wiki pretty much covers all the good stuff, so here is a
"Link"
Diatomaceous earth (

/ˌdaɪ.ətəˌmeɪʃəs ˈɜrθ/) also known as
diatomite or
kieselgur/
kieselguhr, is a naturally occurring, soft, siliceous sedimentary rock that is easily crumbled into a fine white to off-white powder. It has a particle size ranging from less than 1 micrometre to more than 1 millimetre, but typically 10 to 200 micrometres.
[1] This powder has an abrasive feel, similar to pumice powder, and is very light, due to its high porosity. The typical chemical composition of oven dried diatomaceous earth is 80 to 90% silica, with 2 to 4% alumina (attributed mostly to clay minerals) and 0.5 to 2% iron oxide.
[1]Diatomaceous earth consists of fossilized remains of diatoms, a type of hard-shelled algae. It is used as a filtration aid, mild abrasive, mechanical insecticide,
[2] absorbent
for liquids, matting agent for coatings, reinforcing filler in plastics
and rubber, anti-block in plastic films, porous support for chemical
catalysts, cat litter, activator in blood clotting studies, and a stabilizing component of dynamite. As it is also heat-resistant, it can be used as a thermal insulator.
Very interesting properties that make up a really cool picture when the other ingredients are brought into play.

As I remember, diatomite...aka kieselgur...aka diatomacious earth...was a major component in the fabrication of cloth/cordage due to it's mild abrasive properties that helped soften and cleanse the raw material of impurities like oils and foreign/large particles like dirt and bark(which was normally done by physically beating the material or literally stomping it by the workers feet)....as well as it's absorbing properties that allow it to retain whatever compound it is mixed with.
Ok, I'm off to go do stuff, but this topic has me rolling.

Ivo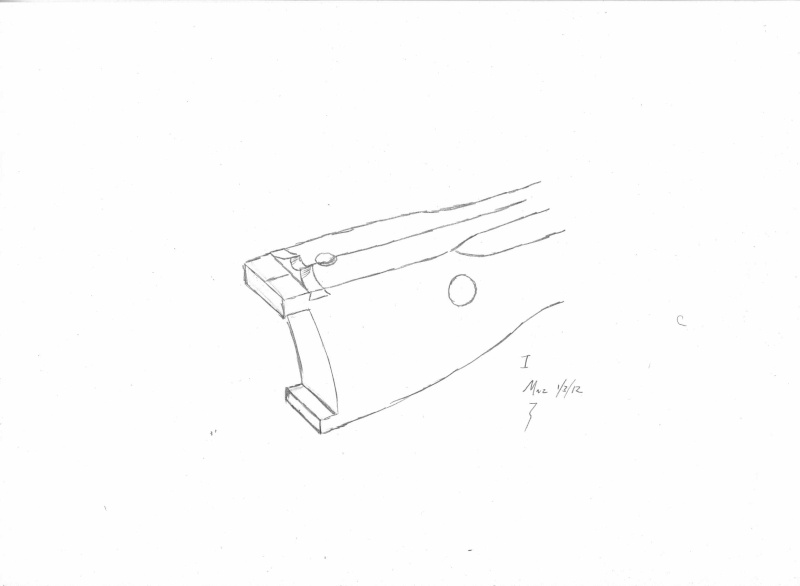 [/img]
[/img]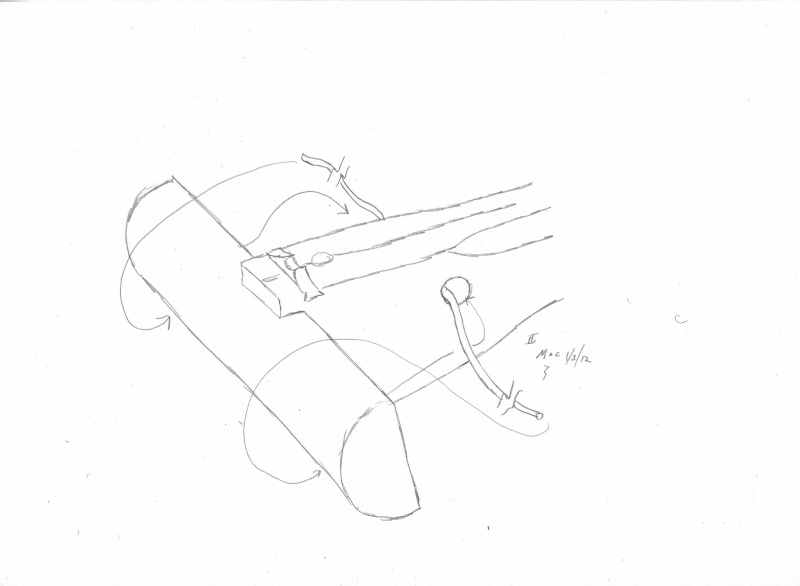 [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img]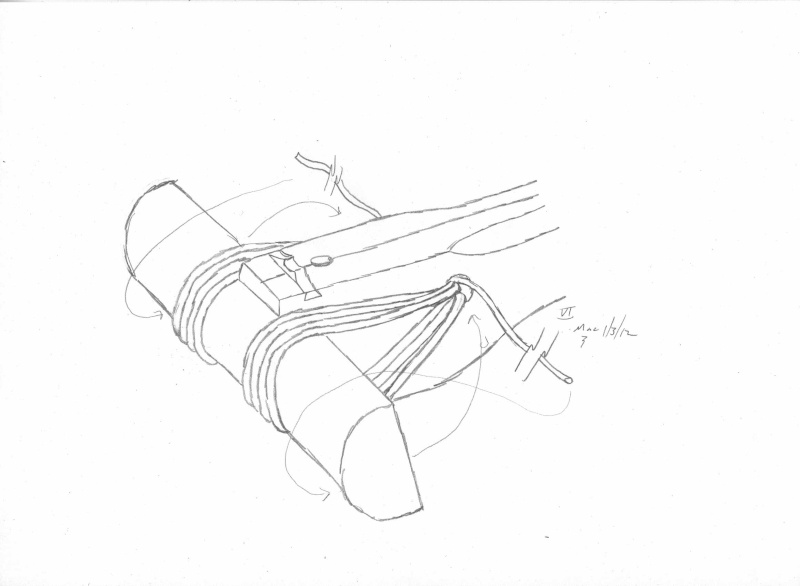 [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img]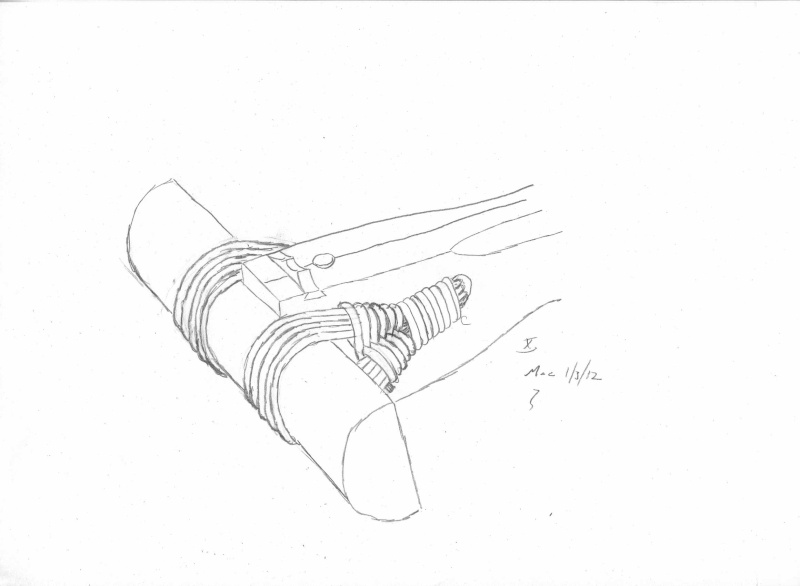 [/img]
[/img]





 [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img] [/img]
[/img] Happens here sometimes...for those who missed this topic, go check it out and save yourself some trouble. >>>
Happens here sometimes...for those who missed this topic, go check it out and save yourself some trouble. >>> 
 ...pretty cool stuff actually.
...pretty cool stuff actually. /ˌdaɪ.ətəˌmeɪʃəs ˈɜrθ/) also known as diatomite or kieselgur/kieselguhr, is a naturally occurring, soft, siliceous sedimentary rock that is easily crumbled into a fine white to off-white powder. It has a particle size ranging from less than 1 micrometre to more than 1 millimetre, but typically 10 to 200 micrometres.[1] This powder has an abrasive feel, similar to pumice powder, and is very light, due to its high porosity. The typical chemical composition of oven dried diatomaceous earth is 80 to 90% silica, with 2 to 4% alumina (attributed mostly to clay minerals) and 0.5 to 2% iron oxide.[1]
/ˌdaɪ.ətəˌmeɪʃəs ˈɜrθ/) also known as diatomite or kieselgur/kieselguhr, is a naturally occurring, soft, siliceous sedimentary rock that is easily crumbled into a fine white to off-white powder. It has a particle size ranging from less than 1 micrometre to more than 1 millimetre, but typically 10 to 200 micrometres.[1] This powder has an abrasive feel, similar to pumice powder, and is very light, due to its high porosity. The typical chemical composition of oven dried diatomaceous earth is 80 to 90% silica, with 2 to 4% alumina (attributed mostly to clay minerals) and 0.5 to 2% iron oxide.[1]


 Private messages
Private messages